Our Research
Brimble Group is at the forefront of pharmaceutical and biotechnology development, with our successes spread over three major research areas. If you are interested in enquiring about research opportunities within the group, head over to the contact page for details.
Harnessing the power of synthetic chemistry to access biomolecules
Our research group comprises a national peptide, peptidomimetic and glycopeptide chemistry facility that occupies a world-class laboratory in the Institute for Innovation in Biotechnology. The peptide synthesis laboratory supports growth in the emerging area of peptide therapeutics that is growing at twice the rate of small molecule therapeutics. This is carried out by engaging with the local biotech community to develop viable drug candidates based on peptide leads.
The following is a representative list of our research projects. Many of our research projects are not listed and we encourage you to contact us should you have further queries.
Boron neutron capture therapy (BNCT)
with Prof John Marshall, Barts Cancer Institute, UK.
Worldwide, there are currently over 17 million people with cancer and in NZ cancer affects more people than all other diseases combined. Innovative therapies such as Boron Neutron Capture Therapy (BNCT) is based upon neutron capture by non-radioactive boron-10 to generate a charged particle in situ. Irradiation of boron-10 results in a nuclear fission reaction generating destructively charged He-4 α-particles, whose energy is confined to a single cell, which restricts lethal damage only to the targeted tumour cell. This project aims to develop peptide boron-10 carriers capable of achieving therapeutic delivery to multiple cancer types thus realising the potential of BNCT.
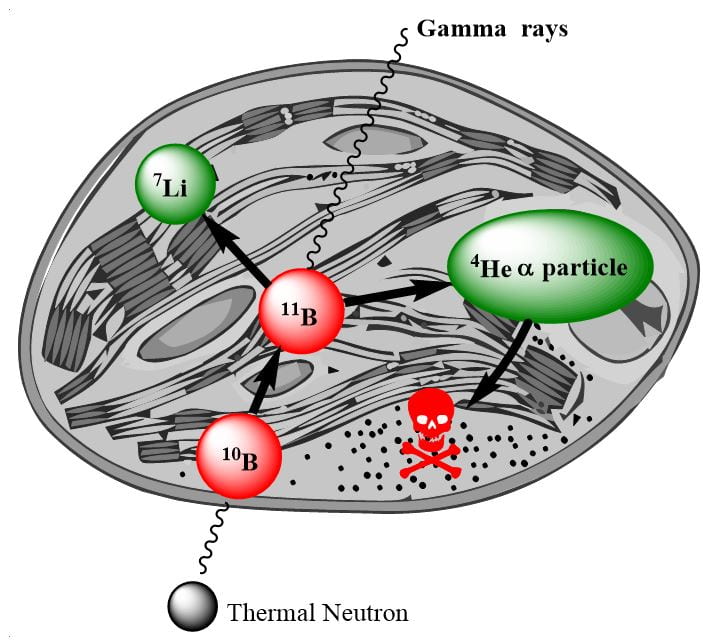
(Above) Basic concept of BNCT, a molecule containing a Boron-10 is localised at the cancer site, captures a thermal neutron producing excited state boron-11 which then undergoes the nuclear fission reaction.
Synthesis of novel peptide antibiotics
with Doherty Institute for Infectious Disease (Melbourne)
Antimicrobial-resistant bacteria has been recognised by the WHO as one of the greatest threats to humankind, and as many as10 million people per annum will succumb to bacterial infections by 2050. Naturally occurring antimicrobial peptides (AMPs), have shown great promise as potential antibiotic candidates. They are especially promising due to the unique and non-specific bactericidal mechanism of action. This project involves the chemical synthesis of naturally-occurring peptides that can then be modified using medicinal chemistry techniques to improve their bioactivity, stability and minimising side effects. Students will become well-versed in chemical synthesis and can evaluate their compounds for activity against Gram-negative and -positive bacteria.
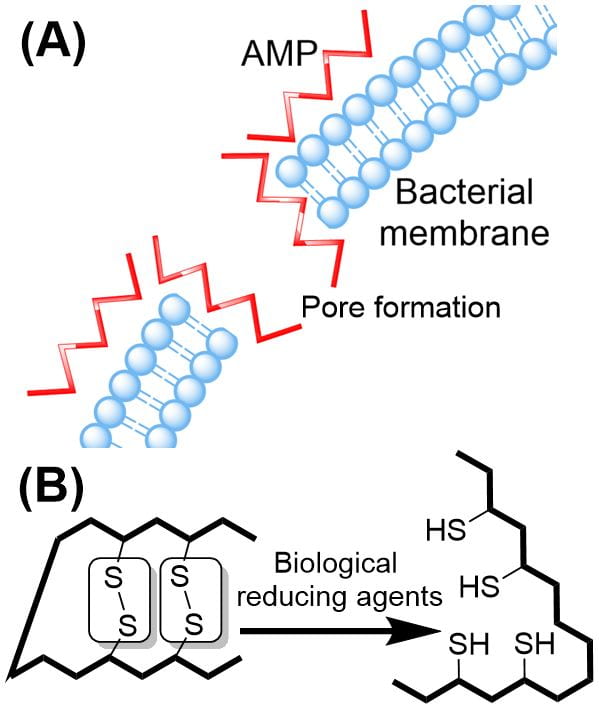
A) Cartoon diagram of an AMP (Red) carpeting and forming pores in a bacterial membrane, leading to cell death. (B) Loss of activity by disulfide bond (bridge) reduction in the body. Replacing the disulphide with novel chemical bridges can restore bioactivity.
Chemical Synthesis of Glycoprotein mimetics
with Dr Lynn Wise (UoO)
Glycoproteins, proteins that are decorated with sugars (glycans) on their surface, are one of the most important protein-based drugs in the 21st century. To be therapeutically useful, the sugars need to be correctly positioned on the protein and changes in glycan coverage can greatly affect the efficacy, half-life, and immunogenicity of proteins. Glycoproteins produced biosynthetically will always afford heterogeneous mixes of beneficial and undesirable glycoforms which compromises their use as therapeutics. This project will employ sophisticated techniques to chemically synthesise proteins, by precisely controlling the location of the glycans, developing a new stratagem in glycoprotein biodrugs.
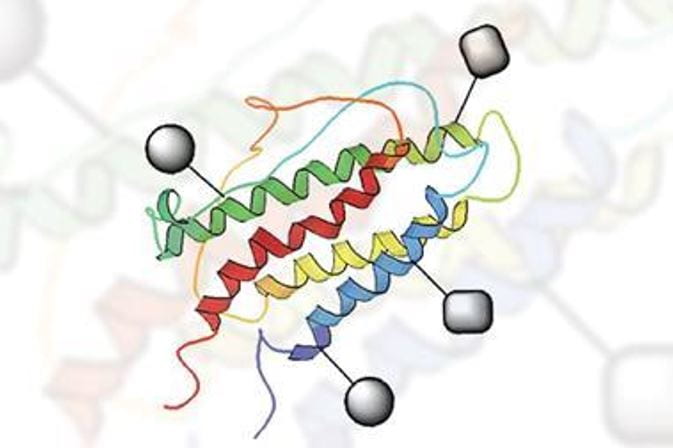
Synthetic EPO prepared by chemical ligation and click chemistry to install synthetic glycans.
Novel chemical staples
Peptides are inherently unconstrained and can adopt multiple conformations, relying on a single conformer to bind to its biological target. The introduction of a chemical “constraint” or a staple can favour one conformer over another and can increase target affinity, cell penetration, and afford protection against enzymatic degradation, thereby realising the potential of peptide as therapeutics. This project will develop novel methodology to constrain antimicrobial peptides via multiple chemical staples and evaluate the peptide constructs using circular dichroism and antibacterial assays.
Antibody Drug conjugates with peptide toxins
with Prof Antony Phillips (SBS)
Antibody drug conjugates (ADCs) e.g. Keytruda, are an emerging FDA-approved cancer therapy that can deliver toxic payloads to cancer cells selectively while minimising toxicity associated with traditional chemotherapy. While the cell-targeting antibodies have been well characterised, the toxins that eradicate the cancers and chemical linker between the toxin and the antibody are less well defined. The project will synthesise innovative self-cleavable linkers and novel peptide-based toxins to determine suitable candidates that can be conjugated to an antibody be assessed in a range of cancer cell lines.
Following Nature’s Lead
Total synthesis
Natural products remain a key source of inspiration for drug discovery and of central importance in understanding small molecule interactions with biomolecules, biosynthetic pathways and pathenogenic organisms. Synthetic access to complex metabolites, and derivative libraries, is an important asset in leveraging their potential as therapeutic lead compounds. Our group employs the continuously-evolving modern synthetic toolkit to develop routes for asymmetric preparation of biological active natural products (examples of completed and current targets are shown below).

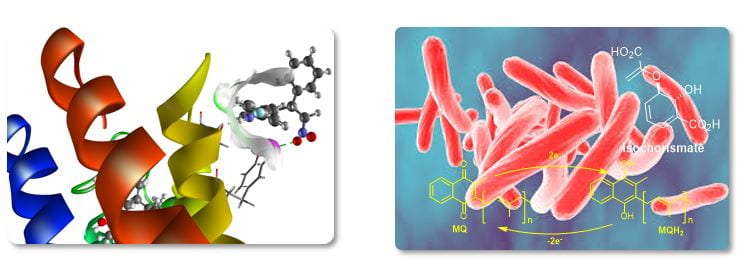
Medicinal chemistry
The tools of organic synthesis are readily applied to the design and preparation of small molecules as potential therapeutics, or as probes to understand the progress of human disease. In collaboration with external groups specialising in molecular or structural biology, microbiology and pharmacology, our group is involved in a range of projects addressing significant health challenges, Currently these include tuberculosis, kauri die-back, marine fouling and CNS disorders (below: left, GPCR ligand design; right, enzyme inhibitors of the causative agent of TB, M. tuberculosis).
Asymmetric method development
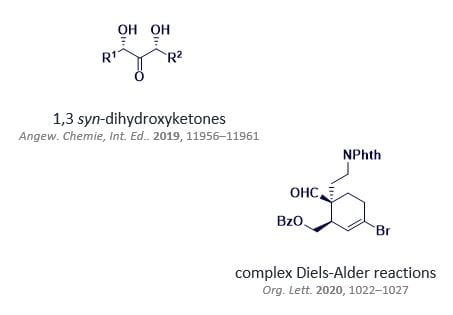
In the course of work towards the synthesis of complex molecules, limitations in current methodology, or occasionally unexpected reactivity, are encountered. Through application of the fundamentals of organic synthesis, our group has been able to develop effective new stereoselective methods to assemble challenging molecular fragments (right, examples of method development towards natural product motifs) and apply these tactically in total synthesis. We remain motivated to identify and pursue similar leads for new reactivity, as the opportunity arises in our synthetic work.
Current Research Projects
The following is a representative list of our research projects. Many of our research projects are not listed and we encourage you to contact us should you have further query.
Natural Product Synthesis: Asymmetric Synthesis of Spiroketals
with A/Prof Dan Furkert
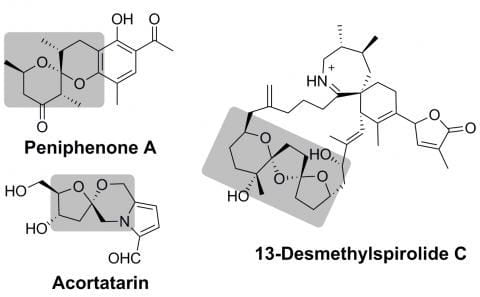
Synthesis of spiroketals and spiroketal-containing natural products is a longstanding interest of our group. These molecular scaffolds, consisting of two (or sometimes even three) rings joined at a quaternary carbon with two bonds to oxygen, are found in a wide range of natural products that demonstrate interesting bioactivity. Some examples of our current and previous targets are shown (spiroketals highlighted in grey).
New Chemical Reactions: Synthetic Applications of Vinyl Azide and Vinyl Amides
with A/Prof Dan Furkert
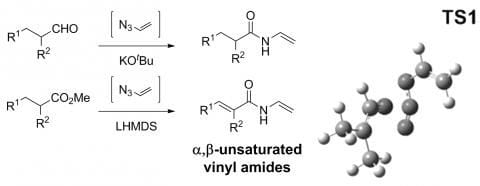
The discovery and development of new reactions offers opportunities to improve synthetic routes to important materials, readily and selectively access previously challenging structures and improve our fundamental understanding of chemical processes. Recently, our group uncovered an unexpected reaction to form alpha,beta-unsaturated vinyl amides directly from esters. Our investigations revealed that the reaction likely involves an unusual [3+2] cylcloaddition of an ester or aldehyde enolate, with in situ generated vinyl azide, a little-used reagent with an intriguing history dating back to original work in 1910.
We are keenly pursuing the possibilities opened up by this new reactivity; for rapid access to previously hard-to-access vinyl amides (versatile synthetic intermediates and useful industrial polymer feedstocks) and synthesis based on them; to explore the mechanistic basis of their reactivity through experiment and calculation (e.g. transition state TS1); and finally to assess the potential of vinyl azide itself in organic synthesis.
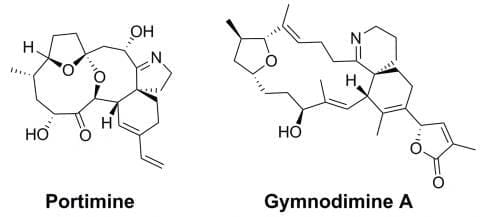
Total Synthesis and Medicinal Chemistry: Spirocyclic Imine Natural Products
with A/Prof Dan Furkert
Shellfish toxins produced by dinoflagellates in during algal blooms such as portimine and gymnodimine are a significant risk to human health – but also provide a stern challenge for existing synthetic methods, and inspirational leads for medicinal chemistry and drug development. Portimine exhibits promising selective anti-cancer activity and apoptosis induction, and gymnodimine is an extremely selective ligand for the nicotinic acetylcholine receptors important in nerve signal transduction. Our group has a strong ongoing interest in the total synthesis of these complex and highly bioactive molecules, and revealing their potential use in medicinal chemistry through structure-activity studies.
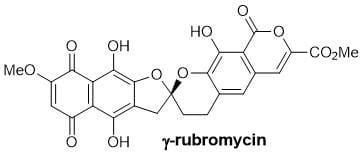
Asymmetric Synthesis of Benzannulated Spiroketals: Towards Novel Telomerase Inhibitors
with A/Prof Dan Furkert
Telomerase inhibitors are of much current interest as a selective approach for the control of human cancer. The rubromycins (inset) are a unique class of antibiotics produced from a strain of Streptomyces that have been shown to inhibit human telomerase. We have previously completed the synthesis of rubromycin, and are currently interested in novel catalytic methods for asymmetric synthesis of this unusual class of compound, that possess a single chiral centre at the spiroketal position.
We will investigate the asymmetric synthesis of benzannulated spiroketals using chiral Lewis acid catalysis, using a new route to prepare the necessary cyclisation substrates only recently identified in our group. In addition to probing the properties and stereochemistry of the compound class, we aim to develop new practical synthetic routes in order to assess the biological activity of chiral lead structures based on rubromycin.
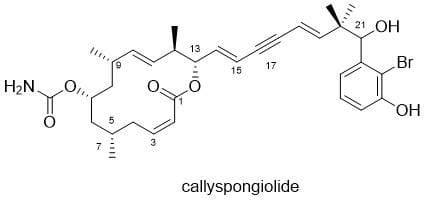
Total Synthesis and Structural Elucidation of Callyspongiolide
with A/Prof Dan Furkert
Callyspongiolide is a 14-membered macrolide isolated in 2013 from an Indonesian marine sponge of the genus Callyspongia Sponges of this genus are known to produce a diverse variety of bioactive secondary metabolites, including polyketides, polyacetylenes, alkaloids and cyclic peptides, but to date, callyspongiolide is the only reported macrolide. Callyspongiolide was found to exhibit potent cytotoxicity against a range of cell lines (L5178Y mouse lymphoma IC50 320 nM, human Jurkat J16 T lymphocytes 70 nM, Ramos B lymphocytes 60 nM). Interestingly, addition of a caspase inhibitor (QVD-OPh) did not attenuate the activity of callyspongiolide, suggesting that it promotes cell death through a caspase independent mechanism.
The relative configuration of the C5, 7, 9 and 12 chiral centres was determined using a combination of 1D NMR proton coupling constants and transannular correlations in the 2D ROESY spectrum. Due to the extremely hindered nature of the secondary alcohol at C21, however, it did not prove possible to prepare any Mosher ester derivatives. As a result, the absolute stereochemistry of callyspongiolide, and the configuration at C21, has not been assigned to date. The C14-19 yn-diene side chain linking the macrolide and bromoaryl domains is unprecedented in macrolide natural products reported to date, although known polyacetylenic algal metabolites are legion.
Total synthesis of the callyspongiolide will enable the complete structural elucidation of the natural product to be completed and permit convenient access to the key sub-structures for SAR investigation of the important biological activity observed.
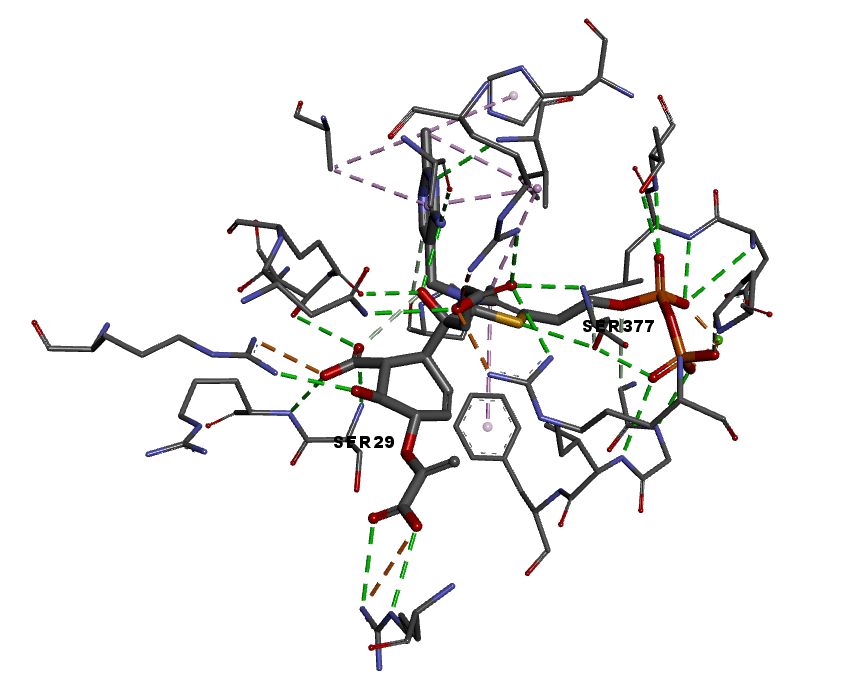
Drug Discovery: Towards New Therapeutics for Mycobacterium tuberculosis (TB)
with A/Prof Shaun Lott, Dr Jodie Johnston and A/Prof Dan Furkert
Tuberculosis (TB) currently affects tens of millions globally and imposes a significant economic and health burden, especially in developing countries. The menaquinone (vitamin K) biosynthetic pathway offers a new drug development target, as control of menaquinone levels can affect the survival of the causative bacterium Mycobacterium tuberculosis in hypoxic environments. (+)‑Isochorismate is the substrate for the first committed step in menaquinone biosynthesis, catalysed by the enzyme MenD.
We are investigating the MenD-catalysed reaction of isochorismate, and its potential as a druggable target in TB. The study draws together complementary expertise in asymmetric synthesis and compound library preparation for SAR study (SCS), alongside structural biology and enzyme kinetics (SBS).
Scientific solutions for the agricultural sector

The primary industry sector (which includes agriculture, forestry and fishing) contributes over half of New Zealand’s total exports (generating NZ$40+ billion in 2019), representing 7% of the country’s GDP.
Agricultural technology, or agritech, is targeted at providing scientific solutions for the agriculture, horticulture, apiculture, aquaculture and fishing sectors, with the goal of improving quality, yield, efficiency, reliability, profitability and sustainability.
Our current research interests range from areas such as animal health/productivity, invasive pest eradication and marine biofouling – through to helping mitigate the environmental footprint left behind by the sector (e.g. reducing greenhouse gas emissions of agricultural origin).
Small molecule inhibitors of biomethanation (methanogenesis) in ruminants

BACKGROUND. Methane concentrations in the atmosphere have more than doubled during the last two centuries, largely as a result of human activities, with agriculture being recognized as the largest source of global methane emissions. Methane absorbs significantly more heat than carbon dioxide, with one tonne of methane being predicted to have the same warming effect as that of 25 tonnes of carbon dioxide. More than 80 percent of New Zealand’s total methane emissions comes from ruminant farm animals – cattle, sheep, goats and deer – mainly as a result of enteric fermentation; the majority of this methane is produced in the rumen, the enlarged modified foregut of ruminant animals. Within the rumen, methanogens are not thought to be essential to the fermentation process of the host animal, but could be considered opportunists, taking advantage of one of the by-products of rumen fermentation: hydrogen gas. This provides an energy source and methanogens combine it with carbon dioxide to produce methane and water. The methane formed is subsequently released into the atmosphere when the animal eructates (belches).
CURRENT PROJECTS. Design and synthesis of small molecule chemical inhibitors which selectively target methane-producing microbes (e.g. methanogens) in the rumen.
SELECTED OUTPUTS
PATENTS: D. Rennison, I.K. Boddy and M.A. Brimble, PCT Number 769755 (filed 10 Nov 2020); D. Rennison, I.K. Boddy and M.A. Brimble, PCT Number 769756 (filed 10 Nov 2020).
Small molecule inhibitors of the nitrification process

Livestock production systems are a major source of greenhouse gases that contribute to climate change. Nitrous oxide (N2O) is a potent greenhouse gas with a long-term global warming potential 298 times that of carbon dioxide (CO2), while nitrate (NO3−) leaching from soil causes water contamination, itself a major environmental issue worldwide. Agriculture is identified as the dominant source for NO3− in surface and ground waters. In grazed grassland systems where animals graze outdoor pastures, most of the N2O and NO3− are from nitrogen (N) returned to the soil in the excreta of the grazing animal, particularly the urine. The use of small molecule nitrification inhibitors (NI) to treat grazed pasture soils in an effort to mitigate NO3− leaching and N2O emissions is widely recognized as one scientifically sound/practically viable solution.

CURRENT PROJECTS. Design and synthesis of small molecule chemical inhibitors which selectively target ammonia-oxidizing bacteria (e.g. nitrosomonas) in the soil.
SELECTED OUTPUTS
PATENTS: TO BE ADDED.
Precision Antimicrobials – Targeted Therapeutics for Food and Companion Animals
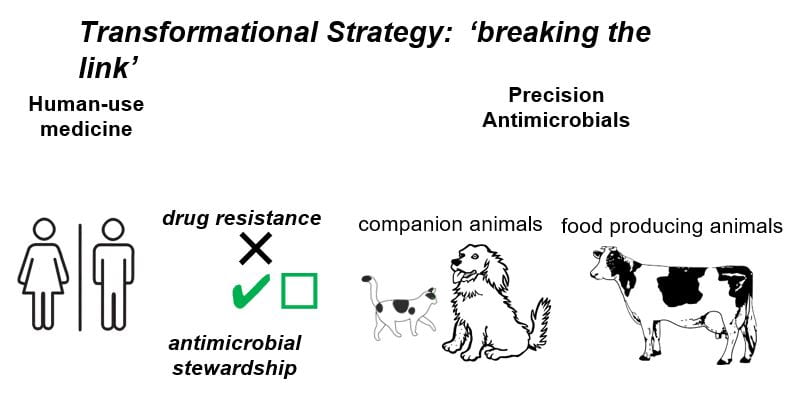
BACKGROUND. Antimicrobial resistance (AMR) is a global threat to public health. Antimicrobial use in animals plays a key role in the emergence, amplification, persistence and transfer of antimicrobial resistance determinants to other ecosystems, including humans. To slow the rise of AMR on farms, the World Health Organisation (WHO) have recommended restricting, or even banning, the use (in animals) of those antibiotics which are considered important for human medicine. Furthermore, these antibiotics (with the exception of ionophores) can no longer be used prophylactically to improve either animal health or productivity.

CURRENT PROJECTS. In an effort to break the link between human and animal medicine through the development of novel animal-only therapeutics we are actively pursuing new antimicrobial compounds, such as ionophores. Ionophores are a class of molecule which has been used safely for decades in the food animal industry. As this class of molecule is not traditionally used in human medicine for the treatment of infectious disease, the opportunity cost to human health is very low. This work represents a transformative therapeutic strategy for the treatment of a targeted range of both drug-sensitive and drug-resistant bacterial infections (Gram-positive and Gram-negative) in food and companion animals.
SELECTED OUTPUTS
C-2 Derivatized sulfonamidoquinolines as antibacterial compounds. E. K. Davison, J. E. McGowan, F. F. Li, A. D. Harper, J. Y. Jeong, S. Mros, N. Harbison-Price, E. M. Van Zuylen, M. K. Knottenbelt, A. Heikal, S. A. Ferguson, M. A. McConnell, G. M. Cook, W. Krittaphol, G. F. Walker, M. A. Brimble and D. Rennison, Bioorg. Med. Chem., 2021, 29, 115837.
Substituted sulfonamide bioisosteres of 8-hydroxyquinoline as zinc-dependent antibacterial compounds. J. E. McGowan, A. D. Harper, E. K. Davison, J. Y. Jeong, S. Mros, N. Harbison-Price, E. M. Van Zuylen, M. K. Knottenbelt, A. Heikal, S. A. Ferguson, M. A. McConnell, G. M. Cook, W. Krittaphol, G. F. Walker, M. A. Brimble and D. Rennison, Bioorg. Med. Chem. Lett., 2020, 30, 127110.
